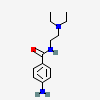
Procainamide
- PROCAINAMIDE
- 51-06-9
- Novocainamide
- Procaine amide
- 4-amino-N-(2-diethylaminoethyl)benzamide
- Create:2005-03-25
- Modify:2025-01-18
 Procainamide Hydrochloride (has salt form).
Procainamide Hydrochloride (has salt form).
- Amide, Procaine
- Apo-Procainamide
- Biocoryl
- Hydrochloride, Procainamide
- Novocainamide
- Novocamid
- Procainamide
- Procainamide Hydrochloride
- Procaine Amide
- Procamide
- Procan
- Procan SR
- Procanbid
- Pronestyl
- Rhythmin
- PROCAINAMIDE
- 51-06-9
- Novocainamide
- Procaine amide
- 4-amino-N-(2-diethylaminoethyl)benzamide
- Biocoryl
- 4-Amino-N-[2-(diethylamino)ethyl]benzamide
- Novocainamid
- Novocamid
- Novocaine amide
- Pronestyl
- Procamide
- Procainamida
- Procainamidum
- Procapan (free base)
- Procan
- p-Aminobenzoic diethylaminoethylamide
- p-Amino-N-(2-diethylaminoethyl)benzamide
- Procainamidum [INN-Latin]
- Procainamida [INN-Spanish]
- NSC 27461
- 4-Amino-N-(2-(diethylamino)ethyl)benzamide
- CHEBI:8428
- Benzamide, 4-amino-N-(2-(diethylamino)ethyl)-
- HSDB 3170
- Sp 100 (pharmaceutical)
- EINECS 200-078-8
- UNII-L39WTC366D
- NSC-27461
- 2-Diethylaminoethylamid kyseliny p-aminobenzoove
- BRN 2214285
- Procainamide (INN)
- L39WTC366D
- DTXSID7023512
- Benzamide, p-amino-N-(2-(diethylamino)ethyl)-
- Benzamide, 4-amino-N-[2-(diethylamino)ethyl]-
- 4-amino-N-(2-diethylaminoethyl) benzamide
- DTXCID403512
- Benzamide, p-amino-N-[2-(diethylamino)ethyl]-
- 4-14-00-01154 (Beilstein Handbook Reference)
- PROC
- NCGC00015859-07
- Rhythmin
- PROCAINAMIDE [INN]
- Procainamidum (INN-Latin)
- Procainamida (INN-Spanish)
- Procanbid; SP 100; SP 100 (pharmaceutical)
- Procainamide [INN:BAN]
- N-(2-(DIETHYLAMINO)ETHYL)-4-AMINOBENZAMIDE
- Amide, Procaine
- CAS-51-06-9
- NSC27461
- CAS-614-39-1
- 2-Diethylaminoethylamid kyseliny p-aminobenzoove [Czech]
- MFCD00066880
- Spectrum_000836
- Maybridge1_004389
- Prestwick0_000337
- Prestwick1_000337
- Prestwick2_000337
- Prestwick3_000337
- Spectrum2_001295
- Spectrum3_000555
- Spectrum4_000487
- Spectrum5_000986
- Lopac-P-9391
- CHEMBL640
- Epitope ID:135397
- PROCAINAMIDE [HSDB]
- Cambridge id 5144127
- PROCAINAMIDE [VANDF]
- Lopac0_000995
- SCHEMBL15914
- BSPBio_000373
- BSPBio_001463
- BSPBio_002229
- CBDivE_003757
- KBioGR_000183
- KBioGR_000973
- KBioSS_000183
- KBioSS_001316
- cid_66068
- BIDD:GT0579
- DivK1c_000931
- PROCAINAMIDE [WHO-DD]
- SPBio_001329
- SPBio_002294
- WLN: ZR DVM2N2&2
- BPBio1_000411
- GTPL4811
- BDBM39344
- HMS553P13
- HY-A0084A
- KBio1_000931
- KBio2_000183
- KBio2_001316
- KBio2_002751
- KBio2_003884
- KBio2_005319
- KBio2_006452
- KBio3_000365
- KBio3_000366
- KBio3_001729
- C01BA02
- NINDS_000931
- Bio1_000391
- Bio1_000880
- Bio1_001369
- Bio2_000183
- Bio2_000663
- HMS1361J05
- HMS1791J05
- HMS1989J05
- HMS2089E13
- HMS3402J05
- Tox21_110246
- STK367963
- AKOS000271131
- Tox21_110246_1
- 1ST9084
- 4-Amino-N-(diethylaminoethyl)benzamide
- CCG-205075
- CS-W009100
- DB01035
- FS-5697
- SDCCGSBI-0050968.P005
- IDI1_000931
- IDI1_033933
- SMP1_000055
- NCGC00015859-01
- NCGC00015859-02
- NCGC00015859-03
- NCGC00015859-04
- NCGC00015859-05
- NCGC00015859-06
- NCGC00015859-08
- NCGC00015859-09
- NCGC00015859-10
- NCGC00015859-11
- NCGC00015859-14
- NCGC00015859-16
- NCGC00015859-18
- NCGC00015859-23
- NCGC00024323-03
- NCGC00024323-04
- NCGC00024323-05
- NCGC00024323-06
- N-(2-Diethylaminoethyl) 4-aminobenzamide
- SBI-0050968.P004
- 4-amino-n-(2-diethylamino-ethyl)-benzamide
- AB00053530
- NS00001509
- EN300-33971
- N1-[2-(diethylamino)ethyl]-4-aminobenzamide
- 4-{N-[2-(diethylamino)ethyl]carbamoyl}aniline
- 4-Amino-N-[2-(diethylamino)ethyl]benzamide #
- C07401
- C75392
- D08421
- AB00053530-13
- AB00053530-15
- AB00053530_16
- L001052
- Q417597
- BRD-K75089421-001-02-5
- BRD-K75089421-003-04-7
- BRD-K75089421-003-05-4
- BRD-K75089421-003-15-3
- BRD-K75089421-003-24-5
- F2173-1035
- 4-azanyl-N-[2-(diethylamino)ethyl]benzamide;hydrochloride
120.04501 100
92.04965 50.50
65.0386 14.90
163.08672 7.20
234.16178 100
92.05049 16.50
206.13076 3.30
161.07185 1.10
234.16127 100
92.05061 20.40
206.13002 6.70
161.07216 3.30
135.0562 2.50
163.08655 100
236.17567 16.10
120.04442 11.50
100.11183 0.70
 Procainamide Hydrochloride (has salt form)
Procainamide Hydrochloride (has salt form)- Cytoplasm
- Membrane

H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral]
H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation]
H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation]
H335 (100%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure; Respiratory tract irritation]
P261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501
(The corresponding statement to each P-code can be found at the GHS Classification page.)
In clinical trials, procainamide was associated with a low rate of serum aminotransferase and alkaline phosphatase elevations. Despite wide scale use, procainamide has only rarely been linked to cases of clinically apparent liver injury. In reported cases, fever and mild symptoms arose within 1 to 3 weeks of starting (or within 1 day of restarting) procainamide, associated with a cholestatic pattern of serum enzyme elevations with mild or no jaundice (Case 1). Immunoallergic features were usually present (fever, rash, leukocytosis). In reported cases, fever resolved immediately and evidence of liver injury within a few days to weeks of stopping procainamide. Liver biopsy may how granulomas in addition to mild nonspecific changes. Interestingly, the hepatotoxicity of procainamide closely resembles that of quinidine, but there is no apparent cross sensitivity to the hepatic injury. In addition, up to 20% of patients on long term procainamide therapy develop autoantibodies, including ANA and LE prep positivity and a proportion develop a “lupus-like” syndrome. These autoimmune conditions, however, typically occur without an accompanying hepatitis, serum enzyme elevations or jaundice.
Likelihood score: C (probable rare cause of clinically apparent liver injury).
M Chen, V Vijay, Q Shi, Z Liu, H Fang, W Tong. FDA-Approved Drug Labeling for the Study of Drug-Induced Liver Injury, Drug Discovery Today, 16(15-16):697-703, 2011. PMID:21624500 DOI:10.1016/j.drudis.2011.05.007
M Chen, A Suzuki, S Thakkar, K Yu, C Hu, W Tong. DILIrank: the largest reference drug list ranked by the risk for developing drug-induced liver injury in humans. Drug Discov Today 2016, 21(4): 648-653. PMID:26948801 DOI:10.1016/j.drudis.2016.02.015
◉ Summary of Use during Lactation
Maternal doses of procainamide 2 grams daily produced low levels of the drug and its active metabolite in the milk of one mother. Although it would not be expected to cause adverse effects in older breastfed infants, the relative lack of data concerning breastfeeding during maternal procainamide therapy would argue for careful monitoring if this drug is used while breastfeeding a neonate possibly Measurement of infant serum levels could help to rule out toxicity if there is a concern.
◉ Effects in Breastfed Infants
Relevant published information was not found as of the revision date.
◉ Effects on Lactation and Breastmilk
Relevant published information was not found as of the revision date.
Patents are available for this chemical structure:
https://patentscope.wipo.int/search/en/result.jsf?inchikey=REQCZEXYDRLIBE-UHFFFAOYSA-N
- Avoid alcohol.
- Take with food. Food reduces irritation.
- BindingDBLICENSEAll data curated by BindingDB staff are provided under the Creative Commons Attribution 3.0 License (https://creativecommons.org/licenses/by/3.0/us/).https://www.bindingdb.org/rwd/bind/info.jsp4-amino-N-[2-(diethylamino)ethyl]benzamide;hydrochloridehttps://www.bindingdb.org/rwd/bind/chemsearch/marvin/MolStructure.jsp?monomerid=39344
- Comparative Toxicogenomics Database (CTD)LICENSEIt is to be used only for research and educational purposes. Any reproduction or use for commercial purpose is prohibited without the prior express written permission of NC State University.http://ctdbase.org/about/legal.jsp
- Drug Gene Interaction database (DGIdb)LICENSEThe data used in DGIdb is all open access and where possible made available as raw data dumps in the downloads section.http://www.dgidb.org/downloadsPROCAINAMIDEhttps://www.dgidb.org/drugs/rxcui:8700
- DrugBankLICENSECreative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode)https://www.drugbank.ca/legal/terms_of_useProcainamidehttps://www.drugbank.ca/drugs/DB01035
- IUPHAR/BPS Guide to PHARMACOLOGYLICENSEThe Guide to PHARMACOLOGY database is licensed under the Open Data Commons Open Database License (ODbL) https://opendatacommons.org/licenses/odbl/. Its contents are licensed under a Creative Commons Attribution-ShareAlike 4.0 International License (http://creativecommons.org/licenses/by-sa/4.0/)https://www.guidetopharmacology.org/about.jsp#licenseGuide to Pharmacology Target Classificationhttps://www.guidetopharmacology.org/targets.jsp
- CAS Common ChemistryLICENSEThe data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc/4.0/
- ChemIDplusProcainamide [INN:BAN]https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000051069ChemIDplus Chemical Information Classificationhttps://pubchem.ncbi.nlm.nih.gov/source/ChemIDplus
- DTP/NCILICENSEUnless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source.https://www.cancer.gov/policies/copyright-reuse
- EPA DSSToxProcainamidehttps://comptox.epa.gov/dashboard/DTXSID7023512CompTox Chemicals Dashboard Chemical Listshttps://comptox.epa.gov/dashboard/chemical-lists/
- European Chemicals Agency (ECHA)LICENSEUse of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.https://echa.europa.eu/web/guest/legal-noticeProcainamide (EC: 200-078-8)https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/104251
- FDA Global Substance Registration System (GSRS)LICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking
- Hazardous Substances Data Bank (HSDB)
- Human Metabolome Database (HMDB)LICENSEHMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications.http://www.hmdb.ca/citingProcainamidehttp://www.hmdb.ca/metabolites/HMDB0015169HMDB0015169_msms_2235739https://hmdb.ca/metabolites/HMDB0015169#spectra
- CCSbaseCCSbase Classificationhttps://ccsbase.net/
- ChEBI
- FDA Pharm ClassesLICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linkingFDA Pharmacological Classificationhttps://www.fda.gov/ForIndustry/DataStandards/StructuredProductLabeling/ucm162549.htm
- LiverTox
- Open TargetsLICENSEDatasets generated by the Open Targets Platform are freely available for download.https://platform-docs.opentargets.org/licencePROCAINAMIDEhttps://platform.opentargets.org/drug/CHEMBL640
- ChEMBLLICENSEAccess to the web interface of ChEMBL is made under the EBI's Terms of Use (http://www.ebi.ac.uk/Information/termsofuse.html). The ChEMBL data is made available on a Creative Commons Attribution-Share Alike 3.0 Unported License (http://creativecommons.org/licenses/by-sa/3.0/).http://www.ebi.ac.uk/Information/termsofuse.htmlChEMBL Protein Target Treehttps://www.ebi.ac.uk/chembl/g/#browse/targets
- ClinicalTrials.govLICENSEThe ClinicalTrials.gov data carry an international copyright outside the United States and its Territories or Possessions. Some ClinicalTrials.gov data may be subject to the copyright of third parties; you should consult these entities for any additional terms of use.https://clinicaltrials.gov/ct2/about-site/terms-conditions#Use
- Therapeutic Target Database (TTD)Procainamidehttps://idrblab.net/ttd/data/drug/details/D0U5SI
- Drug Induced Liver Injury Rank (DILIrank) DatasetLICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking
- NORMAN Suspect List ExchangeLICENSEData: CC-BY 4.0; Code (hosted by ECI, LCSB): Artistic-2.0https://creativecommons.org/licenses/by/4.0/ProcainamideNORMAN Suspect List Exchange Classificationhttps://www.norman-network.com/nds/SLE/
- Drugs and Lactation Database (LactMed)
- Drugs@FDALICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking
- EU Clinical Trials Register
- NIST Mass Spectrometry Data CenterLICENSEData covered by the Standard Reference Data Act of 1968 as amended.https://www.nist.gov/srd/public-lawProcainamidehttp://www.nist.gov/srd/nist1a.cfm
- Japan Chemical Substance Dictionary (Nikkaji)
- KEGGLICENSEAcademic users may freely use the KEGG website. Non-academic use of KEGG generally requires a commercial licensehttps://www.kegg.jp/kegg/legal.htmlAnatomical Therapeutic Chemical (ATC) classificationhttp://www.genome.jp/kegg-bin/get_htext?br08303.kegTarget-based classification of drugshttp://www.genome.jp/kegg-bin/get_htext?br08310.keg
- MassBank of North America (MoNA)LICENSEThe content of the MoNA database is licensed under CC BY 4.0.https://mona.fiehnlab.ucdavis.edu/documentation/license
- Metabolomics Workbench
- NCI Thesaurus (NCIt)LICENSEUnless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source.https://www.cancer.gov/policies/copyright-reuseNCI Thesaurushttps://ncit.nci.nih.gov
- SpectraBaseProcainamidehttps://spectrabase.com/spectrum/9RLCbXRKnOePROCAINAMIDEhttps://spectrabase.com/spectrum/1Xcusc1X24q
- NLM RxNorm TerminologyLICENSEThe RxNorm Terminology is created by the National Library of Medicine (NLM) and is in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from NLM. Credit to the U.S. National Library of Medicine as the source is appreciated but not required. The full RxNorm dataset requires a free license.https://www.nlm.nih.gov/research/umls/rxnorm/docs/termsofservice.htmlprocainamidehttps://rxnav.nlm.nih.gov/id/rxnorm/8700
- PharmGKBLICENSEPharmGKB data are subject to the Creative Commons Attribution-ShareALike 4.0 license (https://creativecommons.org/licenses/by-sa/4.0/).https://www.pharmgkb.org/page/policiesprocainamidehttps://www.pharmgkb.org/chemical/PA451108
- PharosLICENSEData accessed from Pharos and TCRD is publicly available from the primary sources listed above. Please respect their individual licenses regarding proper use and redistribution.https://pharos.nih.gov/aboutprocainamidehttps://pharos.nih.gov/ligands/TM5XNVYPJM31
- Springer Nature
- Thieme ChemistryLICENSEThe Thieme Chemistry contribution within PubChem is provided under a CC-BY-NC-ND 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc-nd/4.0/
- WHO Anatomical Therapeutic Chemical (ATC) ClassificationLICENSEUse of all or parts of the material requires reference to the WHO Collaborating Centre for Drug Statistics Methodology. Copying and distribution for commercial purposes is not allowed. Changing or manipulating the material is not allowed.https://www.whocc.no/copyright_disclaimer/Procainamidehttps://www.whocc.no/atc_ddd_index/?code=C01BA02
- Wikidataprocainamidehttps://www.wikidata.org/wiki/Q417597
- WikipediaProcainamidehttps://en.wikipedia.org/wiki/Procainamide
- Medical Subject Headings (MeSH)LICENSEWorks produced by the U.S. government are not subject to copyright protection in the United States. Any such works found on National Library of Medicine (NLM) Web sites may be freely used or reproduced without permission in the U.S.https://www.nlm.nih.gov/copyright.htmlProcainamidehttps://www.ncbi.nlm.nih.gov/mesh/68011342Voltage-Gated Sodium Channel Blockershttps://www.ncbi.nlm.nih.gov/mesh/68061567Anti-Arrhythmia Agentshttps://www.ncbi.nlm.nih.gov/mesh/68000889
- PubChem
- GHS Classification (UNECE)GHS Classification Treehttp://www.unece.org/trans/danger/publi/ghs/ghs_welcome_e.html
- MolGenieMolGenie Organic Chemistry Ontologyhttps://github.com/MolGenie/ontology/
- PATENTSCOPE (WIPO)SID 403383615https://pubchem.ncbi.nlm.nih.gov/substance/403383615
- NCBI