Oxidopamine Hydrochloride
- C8H11NO3.ClH
- C8H12ClNO3
- Oxidopamine hydrochloride
- 28094-15-7
- 6-Hydroxydopamine hydrochloride
- 5-(2-aminoethyl)benzene-1,2,4-triol hydrochloride
- 6-Hydroxydopamine chloride
- Create:2005-08-08
- Modify:2025-01-18
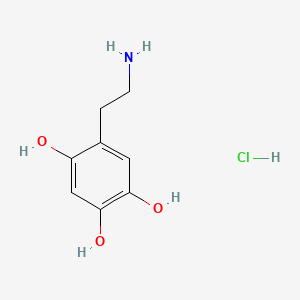
- 6 Hydroxydopamine
- 6-Hydroxydopamine
- 6-OHDA
- Hydrobromide, Oxidopamine
- Hydrochloride, Oxidopamine
- Oxidopamine
- Oxidopamine Hydrobromide
- Oxidopamine Hydrochloride
- Oxidopamine hydrochloride
- 28094-15-7
- 6-Hydroxydopamine hydrochloride
- 5-(2-aminoethyl)benzene-1,2,4-triol hydrochloride
- 6-Hydroxydopamine chloride
- Oxidopamine (hydrochloride)
- 2,4,5-Trihydroxyphenethylamine hydrochloride
- 6-HYDROXYDOPAMINE HCL
- NSC 233898
- UNII-9WBV30HXTL
- 9WBV30HXTL
- EINECS 248-837-2
- DTXSID0045838
- 1,2,3-Benzenetriol, 4-(2-aminoethyl)-, hydrochloride
- 1,2,4-Benzenetriol, 5-(2-aminoethyl)-, hydrochloride
- 3,4,5-Trihydroxyphenethylamine hydrochloride
- 2,5-Dihydroxytyramine hydrochloride
- MFCD00012895
- NSC-233898
- 4-(2-Aminoethyl)-1,2,3-benzenetriol hydrochloride
- 5-(2-aminoethyl)benzene-1,2,4-triol;hydrochloride
- DTXCID8025838
- 6-OHDA
- 6-Hydroxydopamine hydrochloride;6-OHDA hydrochloride
- NCGC00094747-01
- 2-(2,4,5-trihydroxyphenyl)ethan-1-aminium chloride
- Oxidopamine HCl
- 1,2,4-BENZENETRIOL, 5-(2-AMINOETHYL)-, HYDROCHLORIDE (1:1)
- CAS-28094-15-7
- 6-HYDROXYDOPAMINEHYDROCHLORIDE
- 5-(2-Aminoethyl)-1,2,4-benzenetriol hydrochloride
- 5-(2-Aminoethyl)-4-hydroxypyrocatechol hydrochloride
- 6OHDA
- 6Hydroxydopamine chloride
- 6-HD HCl
- Hydrochloride, Oxidopamine
- MLS002207273
- SCHEMBL718426
- SPECTRUM1500450
- 2-(2,4,5-Trihydroxyphenyl)ethylamine hydrochloride
- CHEMBL451197
- 2,5Dihydroxytyraminehydrochloride
- CHEBI:232990
- WLN: Z2R CQ DQ EQ &GH
- HMS1920D14
- Pharmakon1600-01500450
- BCP18603
- HY-B1081
- Tox21_111324
- CCG-40236
- NSC233898
- NSC757259
- AKOS015894730
- Tox21_111324_1
- CS-4648
- NSC-757259
- NCGC00094747-02
- NCGC00167769-04
- 2,4,5Trihydroxyphenethylaminehydrochloride
- 3,5-Trihydroxyphenethylamine hydrochloride
- AS-60772
- SMR001306794
- 2,4,5Trihydroxyphenethylamine hydrochloride
- DB-047336
- NS00083877
- 5(2Aminoethyl)1,2,4benzenetriol hydrochloride
- D87854
- 2(2,4,5 Trihydroxyphenyl)ethylaminehydrochloride
- 5(2Aminoethyl)4hydroxypyrocatechol hydrochloride
- 1,2,4Benzenetriol, 5(2aminoethyl), hydrochloride
- 4-(2-Aminoethyl)-1,3-benzenetriol hydrochloride
- J-016968
- 1,3-Benzenetriol, 4-(2-aminoethyl)-, hydrochloride
- 1,4-Benzenetriol, 5-(2-aminoethyl)-, hydrochloride
- 6-Hydroxydopamine Hydrochloride; 6-OHDA hydrochloride
- Q27273318
- 1,2,4-Benzenetriol,5-(2-aminoethyl)-,hydrochloride(1:1)
- 6-Hydroxydopamine hydrochloride, >=97% (titration), powder

H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation]
H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation]
H335 (100%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure; Respiratory tract irritation]
P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501
(The corresponding statement to each P-code can be found at the GHS Classification page.)
Aggregated GHS information provided per 40 reports by companies from 3 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown.
Skin Irrit. 2 (100%)
Eye Irrit. 2 (100%)
STOT SE 3 (100%)
EYES: First check the victim for contact lenses and remove if present. Flush victim's eyes with water or normal saline solution for 20 to 30 minutes while simultaneously calling a hospital or poison control center. Do not put any ointments, oils, or medication in the victim's eyes without specific instructions from a physician. IMMEDIATELY transport the victim after flushing eyes to a hospital even if no symptoms (such as redness or irritation) develop.
SKIN: IMMEDIATELY flood affected skin with water while removing and isolating all contaminated clothing. Gently wash all affected skin areas thoroughly with soap and water. If symptoms such as redness or irritation develop, IMMEDIATELY call a physician and be prepared to transport the victim to a hospital for treatment.
INHALATION: IMMEDIATELY leave the contaminated area; take deep breaths of fresh air. IMMEDIATELY call a physician and be prepared to transport the victim to a hospital even if no symptoms (such as wheezing, coughing, shortness of breath, or burning in the mouth, throat, or chest) develop. Provide proper respiratory protection to rescuers entering an unknown atmosphere. Whenever possible, Self-Contained Breathing Apparatus (SCBA) should be used; if not available, use a level of protection greater than or equal to that advised under Protective Clothing.
INGESTION: If the victim is conscious and not convulsing, give 1 or 2 glasses of water to dilute the chemical and IMMEDIATELY call a hospital or poison control center. Generally, the induction of vomiting is NOT recommended outside of a physician's care due to the risk of aspirating the chemical into the victim's lungs. However, if the victim is conscious and not convulsing and if medical help is not readily available, consider the risk of inducing vomiting because of the high toxicity of the chemical ingested. Ipecac syrup or salt water may be used in such an emergency. IMMEDIATELY transport the victim to a hospital. If the victim is convulsing or unconscious, do not give anything by mouth, ensure that the victim's airway is open and lay the victim on his/her side with the head lower than the body. DO NOT INDUCE VOMITING. IMMEDIATELY transport the victim to a hospital. (NTP, 1992)
Phenols and Cresols
Salts, Acidic
Patents are available for this chemical structure:
https://patentscope.wipo.int/search/en/result.jsf?inchikey=QLMRJHFAGVFUAC-UHFFFAOYSA-N
- CAMEO ChemicalsLICENSECAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data.https://cameochemicals.noaa.gov/help/reference/terms_and_conditions.htm?d_f=false6-HYDROXYDOPAMINE HYDROCHLORIDEhttps://cameochemicals.noaa.gov/chemical/20499CAMEO Chemical Reactivity Classificationhttps://cameochemicals.noaa.gov/browse/react
- CAS Common ChemistryLICENSEThe data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc/4.0/6-Hydroxydopamine hydrochloridehttps://commonchemistry.cas.org/detail?cas_rn=28094-15-7
- ChemIDplusOxidopamine hydrochloridehttps://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0028094157ChemIDplus Chemical Information Classificationhttps://pubchem.ncbi.nlm.nih.gov/source/ChemIDplus
- DTP/NCILICENSEUnless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source.https://www.cancer.gov/policies/copyright-reuseOxidopamine hydrochloridehttps://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=7572596-Hydroxydopamine hydrochloridehttps://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=233898
- EPA DSSToxOxidopamine hydrochloridehttps://comptox.epa.gov/dashboard/DTXSID0045838CompTox Chemicals Dashboard Chemical Listshttps://comptox.epa.gov/dashboard/chemical-lists/
- European Chemicals Agency (ECHA)LICENSEUse of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.https://echa.europa.eu/web/guest/legal-notice5-(2-aminoethyl)-4-hydroxypyrocatechol hydrochloridehttps://echa.europa.eu/substance-information/-/substanceinfo/100.044.3835-(2-aminoethyl)-4-hydroxypyrocatechol hydrochloride (EC: 248-837-2)https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/2425
- FDA Global Substance Registration System (GSRS)LICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linkingOXIDOPAMINE HYDROCHLORIDEhttps://gsrs.ncats.nih.gov/ginas/app/beta/substances/9WBV30HXTL
- Haz-Map, Information on Hazardous Chemicals and Occupational DiseasesLICENSECopyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission.https://haz-map.com/About6-Hydroxydopamine hydrochloridehttps://haz-map.com/Agents/11295
- ChEMBLLICENSEAccess to the web interface of ChEMBL is made under the EBI's Terms of Use (http://www.ebi.ac.uk/Information/termsofuse.html). The ChEMBL data is made available on a Creative Commons Attribution-Share Alike 3.0 Unported License (http://creativecommons.org/licenses/by-sa/3.0/).http://www.ebi.ac.uk/Information/termsofuse.htmlChEMBL Protein Target Treehttps://www.ebi.ac.uk/chembl/g/#browse/targets
- Drug Gene Interaction database (DGIdb)LICENSEThe data used in DGIdb is all open access and where possible made available as raw data dumps in the downloads section.http://www.dgidb.org/downloadsOXIDOPAMINE HYDROCHLORIDEhttps://www.dgidb.org/drugs/chembl:CHEMBL451197
- SpectraBase5-(2-AMINOETHYL)-1,2,4-BENZENETRIOL, HYDROCHLORIDEhttps://spectrabase.com/spectrum/8TMX8lfKQVJ5-(2-AMINOETHYL)-1,2,4-BENZENETRIOL, HYDROCHLORIDEhttps://spectrabase.com/spectrum/Gbu7Vhl7bya5-(2-AMINOETHYL)-1,2,4-BENZENETRIOL, HYDROCHLORIDEhttps://spectrabase.com/spectrum/DfxyYxgyvwq
- Springer Nature
- Wikidataoxidopamine hydrochloridehttps://www.wikidata.org/wiki/Q27273318
- Medical Subject Headings (MeSH)LICENSEWorks produced by the U.S. government are not subject to copyright protection in the United States. Any such works found on National Library of Medicine (NLM) Web sites may be freely used or reproduced without permission in the U.S.https://www.nlm.nih.gov/copyright.htmlOxidopaminehttps://www.ncbi.nlm.nih.gov/mesh/68016627Adrenergic Agentshttps://www.ncbi.nlm.nih.gov/mesh/68018663Sympatholyticshttps://www.ncbi.nlm.nih.gov/mesh/68013565
- PubChem
- ChEBI
- GHS Classification (UNECE)GHS Classification Treehttp://www.unece.org/trans/danger/publi/ghs/ghs_welcome_e.html
- NORMAN Suspect List ExchangeLICENSEData: CC-BY 4.0; Code (hosted by ECI, LCSB): Artistic-2.0https://creativecommons.org/licenses/by/4.0/NORMAN Suspect List Exchange Classificationhttps://www.norman-network.com/nds/SLE/
- EPA Substance Registry ServicesEPA SRS List Classificationhttps://sor.epa.gov/sor_internet/registry/substreg/LandingPage.do
- MolGenieMolGenie Organic Chemistry Ontologyhttps://github.com/MolGenie/ontology/
- PATENTSCOPE (WIPO)SID 403464677https://pubchem.ncbi.nlm.nih.gov/substance/403464677
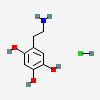

 CID 4624 (Oxidopamine)
CID 4624 (Oxidopamine) CID 313 (Hydrochloric Acid)
CID 313 (Hydrochloric Acid)