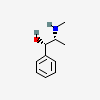d-Ephedrine
- d-Ephedrine
- (+)-Ephedrine
- L-(+)-Ephedrine
- racephedrine
- 321-98-2
- Create:2005-06-24
- Modify:2025-01-11
 Pseudoephedrine (annotation moved to).
Pseudoephedrine (annotation moved to).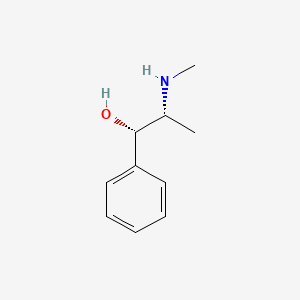
- d-Ephedrine
- (+)-Ephedrine
- L-(+)-Ephedrine
- racephedrine
- 321-98-2
- 90-81-3
- EPHEDRINE, (+)-
- (1S,2R)-Ephedrine
- D-(-)-Ephedrine
- Ephedrine, D-(-)-
- (1S,2R)-2-(methylamino)-1-phenylpropan-1-ol
- NIOSH/KB0650000
- 03VRY66076
- 72087NSY56
- ( inverted exclamation markA)-Ephedrine
- KB06500000
- (+)-Ephedrin
- (+)-(1s,2r)-ephedrine
- Ephedrine DL-form
- (+)-Ephedrine anhydrous
- Benzenemethanol, .alpha.-[(1R)-1-(methylamino)ethyl]-, (.alpha.S)-rel-
- (+-)-Ephedrine
- 2-Methylamino-1-phenylpropan-1-ol
- (1R,2S)-(-)-ephedrine
- EINECS 202-017-0
- EINECS 206-293-3
- Racephedrine [INN:BAN]
- BRN 4231286
- UNII-03VRY66076
- UNII-72087NSY56
- .psi.-Ephedrine
- .psi.-Ephedrin
- BENZENEMETHANOL, .ALPHA.-((1R)-1-(METHYLAMINO)ETHYL)-, (.ALPHA.S)-REL-
- D-.psi.-Ephedrine
- (+/-)-ephedrine
- Novafed (Salt/Mix)
- Sinufed (Salt/Mix)
- BRN 3197916
- Tussaphed (Salt/Mix)
- (+)-erythro-ephedrine
- Symptom 2 (Salt/Mix)
- Benzenemethanol, alpha-((1R)-1-(methylamino)ethyl)-, (alphaS)-
- Benzenemethanol, alpha-(1-(methylamino)ethyl)-, (S-(R*,S*))-
- L(+)-.psi.-Ephedrine
- RACEPHEDRINE [INN]
- .psi.-Ephedrine, (+)-
- (1s,2r)-(-)-ephedrine
- 4-13-00-01881 (Beilstein Handbook Reference)
- RACEPHEDRINE [WHO-DD]
- SCHEMBL420779
- EPHEDRINE, (+-)-
- Benzenemethanol, .alpha.-[1-(methylamino)ethyl]-, [S-(R*,R*)]-
- EPHEDRINE DL-FORM [MI]
- HY-B0980A
- alpha-(1-(Methylaminoethyl)benzenemethanol, (S-(R*,S*)-
- DTXSID20891194
- DTXSID60889333
- Benzenemethanol, .alpha.-[(1R)-1-(methylamino)ethyl]-, (.alpha.S)-
- PDSP1_001345
- PDSP2_001329
- PDSP2_001331
- AKOS027326694
- CS-4471
- 2-(Methylamino)-1-phenyl-1-propanol, d-.psi.- #
- Q22330463
- (R*,S*)-(+-)-alpha-(1-(Methylamino)ethyl)benzyl alcohol
- [S-(R*,R*)]-.alpha.-[1-(methylamino)ethyl]benzenemethanol
- .alpha.-((1S)-1-(Methylamino)ethyl)benzyl alcohol, (.alpha.S)-
- .alpha.-[(1S)-1-(Methylamino)ethyl]benzenemethanol, (.alpha.S)-
- Benzenemethanol, alpha-(1-(methylamino)ethyl)-, (R*,S*)-(+-)-
- BENZENEMETHANOL, .ALPHA.-(1-(METHYLAMINO)ETHYL)-, (S-(R*,S*))-
- Benzenemethanol, alpha-((1R)-1-(methylamino)ethyl)-, (alphaS)-rel-
- BENZENEMETHANOL, .ALPHA.-(1-(METHYLAMINO)ETHYL)-, (R*,S*)-(+/-)-
 Pseudoephedrine (annotation moved to)
Pseudoephedrine (annotation moved to)
H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation]
H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation]
H335 (100%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure; Respiratory tract irritation]
P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501
(The corresponding statement to each P-code can be found at the GHS Classification page.)
Aggregated GHS information provided per 39 reports by companies from 2 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown.
Skin Irrit. 2 (100%)
Eye Irrit. 2 (100%)
STOT SE 3 (100%)
IMAP assessments - Benzenemethanol, .alpha.-[1-(methylamino)ethyl]-, [S-(R*,S*)]-: Environment tier I assessment
IMAP assessments - Benzenemethanol, .alpha.-[1-(methylamino)ethyl]-, [S-(R*,S*)]-: Human health tier I assessment
IMAP assessments - Benzenemethanol, .alpha.-[1-(methylamino)ethyl]-, (R*,S*)-(.+-.)-: Environment tier I assessment
IMAP assessments - Benzenemethanol, .alpha.-[1-(methylamino)ethyl]-, (R*,S*)-(.+-.)-: Human health tier I assessment
Patents are available for this chemical structure:
https://patentscope.wipo.int/search/en/result.jsf?inchikey=KWGRBVOPPLSCSI-PSASIEDQSA-N
- Australian Industrial Chemicals Introduction Scheme (AICIS)Benzenemethanol, .alpha.-[1-(methylamino)ethyl]-, [S-(R*,S*)]-https://services.industrialchemicals.gov.au/search-assessments/
- CAS Common ChemistryLICENSEThe data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc/4.0/
- ChemIDplusRacephedrine [INN:BAN]https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000090813ChemIDplus Chemical Information Classificationhttps://pubchem.ncbi.nlm.nih.gov/source/ChemIDplus
- DrugBankLICENSECreative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode)https://www.drugbank.ca/legal/terms_of_useRacephedrinehttps://www.drugbank.ca/drugs/DB14752
- EPA Chemicals under the TSCABenzenemethanol, .alpha.-[(1R)-1-(methylamino)ethyl]-, (.alpha.S)-rel-https://www.epa.gov/chemicals-under-tscaEPA TSCA Classificationhttps://www.epa.gov/tsca-inventory
- EPA DSSTox(+)-Ephedrinehttps://comptox.epa.gov/dashboard/DTXSID20891194(+/-)-Ephedrinehttps://comptox.epa.gov/dashboard/DTXSID60889333CompTox Chemicals Dashboard Chemical Listshttps://comptox.epa.gov/dashboard/chemical-lists/
- European Chemicals Agency (ECHA)LICENSEUse of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.https://echa.europa.eu/web/guest/legal-notice2-methylamino-1-phenylpropan-1-olhttps://echa.europa.eu/substance-information/-/substanceinfo/100.005.723(R*,S*)-(±)-α-[1-(methylamino)ethyl]benzyl alcoholhttps://echa.europa.eu/substance-information/-/substanceinfo/100.001.8342-methylamino-1-phenylpropan-1-ol (EC: 206-293-3)https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/19724
- FDA Global Substance Registration System (GSRS)LICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking
- ChEMBLLICENSEAccess to the web interface of ChEMBL is made under the EBI's Terms of Use (http://www.ebi.ac.uk/Information/termsofuse.html). The ChEMBL data is made available on a Creative Commons Attribution-Share Alike 3.0 Unported License (http://creativecommons.org/licenses/by-sa/3.0/).http://www.ebi.ac.uk/Information/termsofuse.html
- Crystallography Open Database (COD)LICENSEAll data in the COD and the database itself are dedicated to the public domain and licensed under the CC0 License. Users of the data should acknowledge the original authors of the structural data.https://creativecommons.org/publicdomain/zero/1.0/
- Japan Chemical Substance Dictionary (Nikkaji)
- KEGGLICENSEAcademic users may freely use the KEGG website. Non-academic use of KEGG generally requires a commercial licensehttps://www.kegg.jp/kegg/legal.html
- NCI Thesaurus (NCIt)LICENSEUnless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source.https://www.cancer.gov/policies/copyright-reuseNCI Thesaurushttps://ncit.nci.nih.gov
- NIST Mass Spectrometry Data CenterLICENSEData covered by the Standard Reference Data Act of 1968 as amended.https://www.nist.gov/srd/public-lawPseudoephedrinehttp://www.nist.gov/srd/nist1a.cfm
- SpectraBaseD-EPHEDRINEhttps://spectrabase.com/spectrum/HXXgfuYHiYKD-ephedrinehttps://spectrabase.com/spectrum/7D9lSkOXSOND-EPHEDRINEhttps://spectrabase.com/spectrum/62hgdsBzxuq(+)-Ephedrinehttps://spectrabase.com/spectrum/AKy9MjYTJ9H
- Springer Nature
- Thieme ChemistryLICENSEThe Thieme Chemistry contribution within PubChem is provided under a CC-BY-NC-ND 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc-nd/4.0/
- Wikidata(+)-ephedrinehttps://www.wikidata.org/wiki/Q22330463
- WikipediaRacephedrinehttps://en.wikipedia.org/wiki/Racephedrine
- PubChem
- GHS Classification (UNECE)GHS Classification Treehttp://www.unece.org/trans/danger/publi/ghs/ghs_welcome_e.html
- NORMAN Suspect List ExchangeLICENSEData: CC-BY 4.0; Code (hosted by ECI, LCSB): Artistic-2.0https://creativecommons.org/licenses/by/4.0/NORMAN Suspect List Exchange Classificationhttps://www.norman-network.com/nds/SLE/
- EPA Substance Registry ServicesEPA SRS List Classificationhttps://sor.epa.gov/sor_internet/registry/substreg/LandingPage.do
- MolGenieMolGenie Organic Chemistry Ontologyhttps://github.com/MolGenie/ontology/
- PATENTSCOPE (WIPO)SID 403393641https://pubchem.ncbi.nlm.nih.gov/substance/403393641
- NCBI