Benzyl formate
- Benzyl formate
- 104-57-4
- Benzyl methanoate
- Formic acid, phenylmethyl ester
- Phenylmethyl formate
- Create:2005-03-26
- Modify:2025-02-01
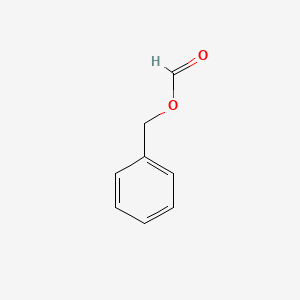
- Benzyl formate
- 104-57-4
- Benzyl methanoate
- Formic acid, phenylmethyl ester
- Phenylmethyl formate
- Benzyl alcohol, formate
- Formic acid, benzyl ester
- Benzyl formiat
- FORMIC ACID BENZYL ESTER
- Ameisensaeurebenzylester
- Benzylester kyseliny mravenci
- FEMA No. 2145
- Carbobenzoxy Group
- NSC 8049
- EINECS 203-214-4
- UNII-79GJF97O0Y
- Benzylester kyseliny mravenci [Czech]
- BRN 2041319
- 79GJF97O0Y
- AI3-02951
- Formic acid-benzyl ester
- NSC-8049
- MFCD00014128
- BENZYL FORMATE [MI]
- BENZYL FORMATE [FCC]
- BENZYL FORMATE [FHFI]
- DTXSID5059298
- FEMA 2145
- 4-06-00-02262 (Beilstein Handbook Reference)
- Benzyl formic acid
- Formate benzyl ester
- (phenylmethyl) methanoate
- carbanic acid benzyl ester
- SCHEMBL503
- WLN: VHO1R
- CHEBI:3057
- DTXCID7032846
- formic acid (phenylmethyl) ester
- NSC8049
- Benzyl formate, analytical standard
- Benzyl formate, natural, 97%, FG
- Benzyl formate, >=95%, FCC, FG
- AKOS015964923
- DB04569
- DS-6364
- BZO
- SY050090
- CS-0128265
- F0050
- NS00012002
- C05613
- D70183
- A801004
- Q4083784
- Formic acid, benzyl ester (6CI,7CI,8CI); Benzyl alcohol, formate; Benzyl formate; Benzyl methanoate; NSC 8049
91.0 99.99
90.0 80.41
136.0 60.54
77.0 32.62
79.0 32.28
91.0 99.99
90.0 89.67
136.0 70.44
79.0 35.81
108.0 34.51
91 999
90 804
136 605
77 326
79 323
91 999
90 897
136 704
79 358
108 345
- Cytoplasm
- Extracellular

P264, P270, P301+P317, P330, and P501
(The corresponding statement to each P-code can be found at the GHS Classification page.)
Aggregated GHS information provided per 1700 reports by companies from 8 notifications to the ECHA C&L Inventory.
Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown.
Patents are available for this chemical structure:
https://patentscope.wipo.int/search/en/result.jsf?inchikey=UYWQUFXKFGHYNT-UHFFFAOYSA-N
- Australian Industrial Chemicals Introduction Scheme (AICIS)Formic acid, phenylmethyl esterhttps://services.industrialchemicals.gov.au/search-assessments/Formic acid, phenylmethyl esterhttps://services.industrialchemicals.gov.au/search-inventory/
- CAS Common ChemistryLICENSEThe data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc/4.0/Benzyl formatehttps://commonchemistry.cas.org/detail?cas_rn=104-57-4
- ChemIDplusChemIDplus Chemical Information Classificationhttps://pubchem.ncbi.nlm.nih.gov/source/ChemIDplus
- DrugBankLICENSECreative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode)https://www.drugbank.ca/legal/terms_of_useBenzyl formatehttps://www.drugbank.ca/drugs/DB04569
- DTP/NCILICENSEUnless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source.https://www.cancer.gov/policies/copyright-reuse
- EPA Chemicals under the TSCAFormic acid, phenylmethyl esterhttps://www.epa.gov/chemicals-under-tscaEPA TSCA Classificationhttps://www.epa.gov/tsca-inventory
- EPA DSSToxFormic acid, phenylmethyl esterhttps://comptox.epa.gov/dashboard/DTXSID5059298CompTox Chemicals Dashboard Chemical Listshttps://comptox.epa.gov/dashboard/chemical-lists/
- European Chemicals Agency (ECHA)LICENSEUse of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.https://echa.europa.eu/web/guest/legal-noticeBenzyl formatehttps://chem.echa.europa.eu/100.002.923Benzyl formate (EC: 203-214-4)https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/98805
- FDA Global Substance Registration System (GSRS)LICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking
- Human Metabolome Database (HMDB)LICENSEHMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications.http://www.hmdb.ca/citingBenzyl formatehttp://www.hmdb.ca/metabolites/HMDB0041485HMDB0041485_cms_28606https://hmdb.ca/metabolites/HMDB0041485#spectra
- International Fragrance Association (IFRA)LICENSE(c) The International Fragrance Association, 2007-2021. All rights reserved.https://ifrafragrance.org/links/copyright
- New Zealand Environmental Protection Authority (EPA)LICENSEThis work is licensed under the Creative Commons Attribution-ShareAlike 4.0 International licence.https://www.epa.govt.nz/about-this-site/general-copyright-statement/
- ChEBI
- LOTUS - the natural products occurrence databaseLICENSEThe code for LOTUS is released under the GNU General Public License v3.0.https://lotus.nprod.net/Benzyl formatehttps://www.wikidata.org/wiki/Q4083784LOTUS Treehttps://lotus.naturalproducts.net/
- EPA Chemical and Products Database (CPDat)Formic acid, phenylmethyl esterhttps://comptox.epa.gov/dashboard/DTXSID5059298#exposureEPA CPDat Classificationhttps://www.epa.gov/chemical-research/chemical-and-products-database-cpdat
- EU Food Improvement Agents
- Joint FAO/WHO Expert Committee on Food Additives (JECFA)LICENSEPermission from WHO is not required for the use of WHO materials issued under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Intergovernmental Organization (CC BY-NC-SA 3.0 IGO) licence.https://www.who.int/about/policies/publishing/copyright
- FDA Substances Added to FoodLICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking
- Flavor and Extract Manufacturers Association (FEMA)BENZYL FORMATEhttps://www.femaflavor.org/flavor-library/benzyl-formate
- FooDBLICENSEFooDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (FooDB) and the original publication.https://foodb.ca/aboutBenzyl formatehttps://foodb.ca/compounds/FDB021450
- MassBank of North America (MoNA)LICENSEThe content of the MoNA database is licensed under CC BY 4.0.https://mona.fiehnlab.ucdavis.edu/documentation/license
- NIST Mass Spectrometry Data CenterLICENSEData covered by the Standard Reference Data Act of 1968 as amended.https://www.nist.gov/srd/public-lawFormic acid, phenylmethyl esterhttp://www.nist.gov/srd/nist1a.cfm
- SpectraBaseFORMIC ACID, PHENYLMETHYL ESTERhttps://spectrabase.com/spectrum/IEHUc9eOGpoFORMIC ACID, PHENYLMETHYL ESTERhttps://spectrabase.com/spectrum/JB7arVV7IMVBenzyl formatehttps://spectrabase.com/spectrum/8VsnEgddby2Benzyl formatehttps://spectrabase.com/spectrum/5sbWuBxtvrZBenzyl formatehttps://spectrabase.com/spectrum/B4XupTHVBTGBenzyl formatehttps://spectrabase.com/spectrum/BFIBcXrThXJBenzyl formatehttps://spectrabase.com/spectrum/HrffKPFJS1zFormic acid, benzyl esterhttps://spectrabase.com/spectrum/29qCv59StnfBenzyl formatehttps://spectrabase.com/spectrum/mbiNMkTziCS-CIS-CONFORMATIONhttps://spectrabase.com/spectrum/AsgPoaKnjLfBenzyl formatehttps://spectrabase.com/spectrum/IKHB8sog0v0Benzyl formatehttps://spectrabase.com/spectrum/LaLCGradQlkBenzyl formatehttps://spectrabase.com/spectrum/93kWsMRI9u2
- Japan Chemical Substance Dictionary (Nikkaji)
- KEGGLICENSEAcademic users may freely use the KEGG website. Non-academic use of KEGG generally requires a commercial licensehttps://www.kegg.jp/kegg/legal.html
- KNApSAcK Species-Metabolite Database
- Natural Product Activity and Species Source (NPASS)
- MassBank Europe
- Metabolomics Workbench
- NMRShiftDB
- NORMAN Suspect List ExchangeLICENSEData: CC-BY 4.0; Code (hosted by ECI, LCSB): Artistic-2.0https://creativecommons.org/licenses/by/4.0/Formic acid, phenylmethyl esterNORMAN Suspect List Exchange Classificationhttps://www.norman-network.com/nds/SLE/
- Springer Nature
- SpringerMaterialsformic acid benzyl esterhttps://materials.springer.com/substanceprofile/docs/smsid_ofqblmerqtjmxylr
- Thieme ChemistryLICENSEThe Thieme Chemistry contribution within PubChem is provided under a CC-BY-NC-ND 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc-nd/4.0/
- Wikidatabenzyl formatehttps://www.wikidata.org/wiki/Q4083784
- PubChem
- Medical Subject Headings (MeSH)LICENSEWorks produced by the U.S. government are not subject to copyright protection in the United States. Any such works found on National Library of Medicine (NLM) Web sites may be freely used or reproduced without permission in the U.S.https://www.nlm.nih.gov/copyright.htmlbenzyl formatehttps://www.ncbi.nlm.nih.gov/mesh/2027568
- GHS Classification (UNECE)GHS Classification Treehttp://www.unece.org/trans/danger/publi/ghs/ghs_welcome_e.html
- EPA Substance Registry ServicesEPA SRS List Classificationhttps://sor.epa.gov/sor_internet/registry/substreg/LandingPage.do
- MolGenieMolGenie Organic Chemistry Ontologyhttps://github.com/MolGenie/ontology/
- PATENTSCOPE (WIPO)SID 403385619https://pubchem.ncbi.nlm.nih.gov/substance/403385619





