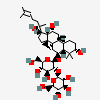
Ginsenoside Rf
- Ginsenoside Rf
- 52286-58-5
- Panaxoside RF
- ginsenoside-rf
- JOS8BON5YW
- Create:2005-06-24
- Modify:2025-01-18
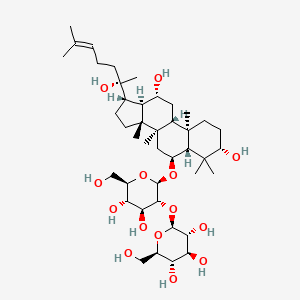
- Ginsenoside Rf
- 52286-58-5
- Panaxoside RF
- ginsenoside-rf
- JOS8BON5YW
- UNII-JOS8BON5YW
- CHEBI:67986
- MFCD00210509
- (2S,3R,4S,5S,6R)-2-[(2R,3R,4S,5S,6R)-2-[[(3S,5R,6S,8R,9R,10R,12R,13R,14R,17S)-3,12-dihydroxy-17-[(2S)-2-hydroxy-6-methylhept-5-en-2-yl]-4,4,8,10,14-pentamethyl-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-6-yl]oxy]-4,5-dihydroxy-6-(hydroxymethyl)oxan-3-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol
- DTXSID90904205
- GINSENOSIDE- RF
- GinsenosideRf
- (beta,6alpha,12beta)-3,12,20-trihydroxydammar-24-en-6-yl 2-O-beta-D-glucopyranosyl-beta-D-glucopyranoside
- CHEMBL1773987
- ss-D-Glucopyranoside, (3ss,6a,12ss)-3,12,20-trihydroxydammar-24-en-6-yl 2-O-ss-D-glucopyranosyl-; Dammarane, ss-D-glucopyranoside deriv.; (3ss,6a,12ss)-3,12,20-Trihydroxydammar-24-en-6-yl 2-O-ss-D-glucopyranosyl-ss-D-glucopyranoside; Ginsenoside Rf; Panaxoside Rf
- Ginsenoside Rf (Standard)
- SCHEMBL22607970
- HY-N0601R
- DTXCID101333354
- Ginsenoside Rf, analytical standard
- HY-N0601
- AKOS027251120
- CS-3841
- 1ST40274
- AS-56543
- beta-D-Glucopyranoside, (3beta,6alpha,12beta)-3,12,20-trihydroxydammar-24-en-6-yl 2-O-beta-D-glucopyranosyl-
- DA-63765
- C08945
- Q-100589
- Q27104950
- Ginsenoside Rf, primary pharmaceutical reference standard
- GINSENOSIDE RF (CONSTITUENT OF AMERICAN GINSENG, ASIAN GINSENG, AND TIENCHI GINSENG)
- (3beta,6alpha,12beta)-3,12,20-Trihydroxydammar-24-en-6-yl 2-O-beta-D-glucopyranosyl-beta-D-glucopyranoside
- .BETA.-D-GLUCOPYRANOSIDE, (3.BETA.,6.ALPHA.,12.BETA.)-3,12,20-TRIHYDROXYDAMMAR-24-EN-6-YL 2-O-.BETA.-D-GLUCOPYRANOSYL-
- GINSENOSIDE RF (CONSTITUENT OF AMERICAN GINSENG, ASIAN GINSENG, AND TIENCHI GINSENG) [DSC]
638 100
476 100

P264, P270, P301+P317, P330, and P501
(The corresponding statement to each P-code can be found at the GHS Classification page.)
Aggregated GHS information provided per 41 reports by companies from 2 notifications to the ECHA C&L Inventory.
Reported as not meeting GHS hazard criteria per 1 of 41 reports by companies. For more detailed information, please visit ECHA C&L website.
There is 1 notification provided by 40 of 41 reports by companies with hazard statement code(s).
Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown.
Patents are available for this chemical structure:
https://patentscope.wipo.int/search/en/result.jsf?inchikey=UZIOUZHBUYLDHW-XUBRWZAZSA-N
- CAS Common ChemistryLICENSEThe data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc/4.0/Ginsenoside Rfhttps://commonchemistry.cas.org/detail?cas_rn=52286-58-5
- ChemIDplusChemIDplus Chemical Information Classificationhttps://pubchem.ncbi.nlm.nih.gov/source/ChemIDplus
- EPA DSSToxGinsenoside Rfhttps://comptox.epa.gov/dashboard/DTXSID90904205CompTox Chemicals Dashboard Chemical Listshttps://comptox.epa.gov/dashboard/chemical-lists/
- European Chemicals Agency (ECHA)LICENSEUse of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.https://echa.europa.eu/web/guest/legal-noticeGinsenoside RF (EC: 634-870-3)https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/167632
- FDA Global Substance Registration System (GSRS)LICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking
- ChEBI
- LOTUS - the natural products occurrence databaseLICENSEThe code for LOTUS is released under the GNU General Public License v3.0.https://lotus.nprod.net/Ginsenoside Rfhttps://www.wikidata.org/wiki/Q27104950LOTUS Treehttps://lotus.naturalproducts.net/
- ChEMBLLICENSEAccess to the web interface of ChEMBL is made under the EBI's Terms of Use (http://www.ebi.ac.uk/Information/termsofuse.html). The ChEMBL data is made available on a Creative Commons Attribution-Share Alike 3.0 Unported License (http://creativecommons.org/licenses/by-sa/3.0/).http://www.ebi.ac.uk/Information/termsofuse.html
- Comparative Toxicogenomics Database (CTD)LICENSEIt is to be used only for research and educational purposes. Any reproduction or use for commercial purpose is prohibited without the prior express written permission of NC State University.http://ctdbase.org/about/legal.jspginsenoside Rfhttps://ctdbase.org/detail.go?type=chem&acc=C055328
- Japan Chemical Substance Dictionary (Nikkaji)
- KEGGLICENSEAcademic users may freely use the KEGG website. Non-academic use of KEGG generally requires a commercial licensehttps://www.kegg.jp/kegg/legal.htmlPhytochemical compoundshttp://www.genome.jp/kegg-bin/get_htext?br08003.keg
- Natural Product Activity and Species Source (NPASS)
- MassBank of North America (MoNA)LICENSEThe content of the MoNA database is licensed under CC BY 4.0.https://mona.fiehnlab.ucdavis.edu/documentation/license
- Metabolomics Workbench
- Springer Nature
- Wikidataginsenoside Rfhttps://www.wikidata.org/wiki/Q27104950
- PubChem
- Medical Subject Headings (MeSH)LICENSEWorks produced by the U.S. government are not subject to copyright protection in the United States. Any such works found on National Library of Medicine (NLM) Web sites may be freely used or reproduced without permission in the U.S.https://www.nlm.nih.gov/copyright.htmlginsenoside Rfhttps://www.ncbi.nlm.nih.gov/mesh/67055328
- GHS Classification (UNECE)GHS Classification Treehttp://www.unece.org/trans/danger/publi/ghs/ghs_welcome_e.html
- MolGenieMolGenie Organic Chemistry Ontologyhttps://github.com/MolGenie/ontology/
- PATENTSCOPE (WIPO)SID 388579651https://pubchem.ncbi.nlm.nih.gov/substance/388579651