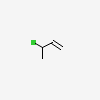
3-Chloro-1-butene
- 3-CHLORO-1-BUTENE
- 563-52-0
- 1-Butene, 3-chloro-
- 3-Chlorobut-1-ene
- 1-Methylallyl chloride
- Create:2005-03-26
- Modify:2025-01-11
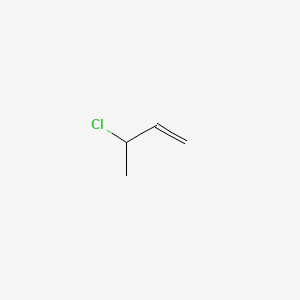
- 3-CHLORO-1-BUTENE
- 563-52-0
- 1-Butene, 3-chloro-
- 3-Chlorobut-1-ene
- 1-Methylallyl chloride
- .alpha.-Methallyl chloride
- alpha-Methallyl chloride
- alpha-Methylallyl chloride
- gamma-Chloro-alpha-butylene
- 2-Chloro-3-butene
- .alpha.-Methylallyl chloride
- .gamma.-Chloro-.alpha.-butylene
- EINECS 209-252-8
- NSC 60192
- UNII-58S8R4439O
- NSC-60192
- 58S8R4439O
- DTXSID50870614
- 3-CHLORO-1-BUTENE [MI]
- DL-.ALPHA.-METHALLYL CHLORIDE
- (+/-)-3-CHLORO-1-BUTENE
- 3-CHLORO-1-BUTENE DL-FORM [MI]
- 3Chlorobut1ene
- Methylallylchlorid
- 1Butene, 3chloro
- MFCD00000869
- ?-Chloro-a-butylene
- 1Methylallyl chloride
- a-Methylallyl chloride
- alphaMethallyl chloride
- gammaChloroalphabutylene
- dl-a-Methallyl chloride
- alphaMethylallyl chloride
- 1-Methyl-1-chloro-2-propene
- DTXCID50818319
- DL-ALPHA-METHALLYL CHLORIDE
- 3-CHLORO-1-BUTENE DL-FORM
- NSC60192
- AKOS009157277
- DB-052898
- NS00042356
- EN300-49169
- F11733
- Q27261614
- 3-Chloro-1-butene, contains sodium/potassium carbonate as stabilizer, 97%



H225 (53.8%): Highly Flammable liquid and vapor [Danger Flammable liquids]
H302 (47.5%): Harmful if swallowed [Warning Acute toxicity, oral]
H314 (46.2%): Causes severe skin burns and eye damage [Danger Skin corrosion/irritation]
H317 (97.5%): May cause an allergic skin reaction [Warning Sensitization, Skin]
H332 (46.2%): Harmful if inhaled [Warning Acute toxicity, inhalation]
H412 (46.2%): Harmful to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard]
P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P272, P273, P280, P301+P317, P301+P330+P331, P302+P352, P302+P361+P354, P303+P361+P353, P304+P340, P305+P354+P338, P316, P317, P321, P330, P333+P317, P362+P364, P363, P370+P378, P403+P235, P405, and P501
(The corresponding statement to each P-code can be found at the GHS Classification page.)
Aggregated GHS information provided per 80 reports by companies from 4 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown.
Flam. Liq. 2 (53.8%)
Acute Tox. 4 (47.5%)
Skin Corr. 1B (46.2%)
Skin Sens. 1 (97.5%)
Acute Tox. 4 (46.2%)
Aquatic Chronic 3 (46.2%)
Patents are available for this chemical structure:
https://patentscope.wipo.int/search/en/result.jsf?inchikey=VZGLVCFVUREVDP-UHFFFAOYSA-N
- CAS Common ChemistryLICENSEThe data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc/4.0/3-Chloro-1-butenehttps://commonchemistry.cas.org/detail?cas_rn=563-52-0
- ChemIDplusChemIDplus Chemical Information Classificationhttps://pubchem.ncbi.nlm.nih.gov/source/ChemIDplus
- DTP/NCILICENSEUnless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source.https://www.cancer.gov/policies/copyright-reuse
- EPA Chemicals under the TSCA1-Butene, 3-chloro-https://www.epa.gov/chemicals-under-tscaEPA TSCA Classificationhttps://www.epa.gov/tsca-inventory
- EPA DSSTox1-Butene, 3-chloro-https://comptox.epa.gov/dashboard/DTXSID50870614CompTox Chemicals Dashboard Chemical Listshttps://comptox.epa.gov/dashboard/chemical-lists/
- European Chemicals Agency (ECHA)LICENSEUse of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.https://echa.europa.eu/web/guest/legal-notice3-chlorobut-1-ene (EC: 209-252-8)https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/45355
- FDA Global Substance Registration System (GSRS)LICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking3-CHLORO-1-BUTENEhttps://gsrs.ncats.nih.gov/ginas/app/beta/substances/58S8R4439O
- New Zealand Environmental Protection Authority (EPA)LICENSEThis work is licensed under the Creative Commons Attribution-ShareAlike 4.0 International licence.https://www.epa.govt.nz/about-this-site/general-copyright-statement/
- Haz-Map, Information on Hazardous Chemicals and Occupational DiseasesLICENSECopyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission.https://haz-map.com/About3-Chloro-1-butenehttps://haz-map.com/Agents/19339
- Japan Chemical Substance Dictionary (Nikkaji)
- NIST Mass Spectrometry Data CenterLICENSEData covered by the Standard Reference Data Act of 1968 as amended.https://www.nist.gov/srd/public-law1-Butene, 3-chloro-http://www.nist.gov/srd/nist1a.cfm
- SpectraBase1-Butene, 3-chloro-https://spectrabase.com/spectrum/G5kJSzKfy7d1-Butene, 3-chloro-https://spectrabase.com/spectrum/I133p9sNRb93-CHLORO-1-BUTENEhttps://spectrabase.com/spectrum/5nnQEzsga203-chloro-1-butenehttps://spectrabase.com/spectrum/EtM5K3H0XZh3-CHLORO-1-BUTENEhttps://spectrabase.com/spectrum/FSOYN9bpRUm3-Chloro-1-butenehttps://spectrabase.com/spectrum/izD7ZLy3bP3-CHLORO-1-BUTENEhttps://spectrabase.com/spectrum/IBfp04zhfLm3-Chloro-1-butenehttps://spectrabase.com/spectrum/CgpmL7y86h51-BUTENE, 3-CHLORO-,https://spectrabase.com/spectrum/3iA5YJFuHN43-Chloro-1-butenehttps://spectrabase.com/spectrum/K3NceiB8F7K1-Butene, 3-chloro-https://spectrabase.com/spectrum/BzCXB1W5KFa1-Butene, 3-chloro-https://spectrabase.com/spectrum/J6lh9RD4V5C3-Chloro-1-butenehttps://spectrabase.com/spectrum/7ckH5H1SXh63-Chloro-1-butenehttps://spectrabase.com/spectrum/6AWrvwzwODA3-Chloro-1-butenehttps://spectrabase.com/spectrum/GHUneP4GxpS
- Springer Nature
- Thieme ChemistryLICENSEThe Thieme Chemistry contribution within PubChem is provided under a CC-BY-NC-ND 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc-nd/4.0/
- Wikidata3-Chloro-1-butenehttps://www.wikidata.org/wiki/Q27261614
- Wiley
- PubChem
- Medical Subject Headings (MeSH)LICENSEWorks produced by the U.S. government are not subject to copyright protection in the United States. Any such works found on National Library of Medicine (NLM) Web sites may be freely used or reproduced without permission in the U.S.https://www.nlm.nih.gov/copyright.html3-chloro-1-butenehttps://www.ncbi.nlm.nih.gov/mesh/67525991
- GHS Classification (UNECE)GHS Classification Treehttp://www.unece.org/trans/danger/publi/ghs/ghs_welcome_e.html
- NORMAN Suspect List ExchangeLICENSEData: CC-BY 4.0; Code (hosted by ECI, LCSB): Artistic-2.0https://creativecommons.org/licenses/by/4.0/NORMAN Suspect List Exchange Classificationhttps://www.norman-network.com/nds/SLE/
- EPA Substance Registry ServicesEPA SRS List Classificationhttps://sor.epa.gov/sor_internet/registry/substreg/LandingPage.do
- MolGenieMolGenie Organic Chemistry Ontologyhttps://github.com/MolGenie/ontology/
- PATENTSCOPE (WIPO)SID 403408427https://pubchem.ncbi.nlm.nih.gov/substance/403408427