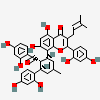
Kuwanon G
PubChem CID
5281667
Molecular Formula
Synonyms
- Kuwanon G
- 75629-19-5
- moracenin b
- Kuwanone G
- KuwanonG
Molecular Weight
692.7 g/mol
Computed by PubChem 2.2 (PubChem release 2021.10.14)
Dates
- Create:2005-06-24
- Modify:2025-01-18
Description
Kuwanone G is a tetrahydroxyflavone isolated from the root barks of Morus alba and has been shown to exhibit anti-inflammatory activity. It has a role as an anti-inflammatory agent and a plant metabolite. It is a tetrahydroxyflavone and a member of resorcinols.
Chemical Structure Depiction
Conformer generation is disallowed since too many atoms
8-[(1S,5R,6S)-6-(2,4-dihydroxybenzoyl)-5-(2,4-dihydroxyphenyl)-3-methylcyclohex-2-en-1-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methylbut-2-enyl)chromen-4-one
Computed by Lexichem TK 2.7.0 (PubChem release 2021.10.14)
InChI=1S/C40H36O11/c1-18(2)4-8-26-38(50)36-33(48)17-32(47)35(40(36)51-39(26)25-11-7-22(43)16-31(25)46)28-13-19(3)12-27(23-9-5-20(41)14-29(23)44)34(28)37(49)24-10-6-21(42)15-30(24)45/h4-7,9-11,13-17,27-28,34,41-48H,8,12H2,1-3H3/t27-,28-,34-/m0/s1
Computed by InChI 1.0.6 (PubChem release 2021.10.14)
APPXYONGBIXGRO-AIQWNVMPSA-N
Computed by InChI 1.0.6 (PubChem release 2021.10.14)
CC1=C[C@@H]([C@H]([C@@H](C1)C2=C(C=C(C=C2)O)O)C(=O)C3=C(C=C(C=C3)O)O)C4=C(C=C(C5=C4OC(=C(C5=O)CC=C(C)C)C6=C(C=C(C=C6)O)O)O)O
Computed by OEChem 2.3.0 (PubChem release 2024.12.12)
C40H36O11
Computed by PubChem 2.2 (PubChem release 2021.10.14)
kuwanon G
- Kuwanon G
- 75629-19-5
- moracenin b
- Kuwanone G
- KuwanonG
- kumanon G
- CHEBI:6146
- GQ6QVK8YZM
- 8-[(1S,5R,6S)-6-(2,4-dihydroxybenzoyl)-5-(2,4-dihydroxyphenyl)-3-methylcyclohex-2-en-1-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methylbut-2-enyl)chromen-4-one
- Kuwanon
- 4H-1-Benzopyran-4-one, 8-[(1S,5R,6S)-6-(2,4-dihydroxybenzoyl)-5-(2,4-dihydroxyphenyl)-3-methyl-2-cyclohexen-1-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methyl-2-butenyl)-
- 8-[(1S,6S)-6-(2,4-dihydroxybenzoyl)-5-(2,4-dihydroxyphenyl)-3-methylcyclohex-2-en-1-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methylbut-2-enyl)chromen-4-one
- NSC-356888
- Kuwanon G (Standard)
- UNII-GQ6QVK8YZM
- CHEMBL444942
- SCHEMBL20211451
- HY-N4247R
- DTXSID40226540
- APPXYONGBIXGRO-AIQWNVMPSA-N
- 8-[(1S,5R,6S)-6-(2,4-dihydroxybenzoyl)-5-(2,4-dihydroxyphenyl)-3-methyl-cyclohex-2-en-1-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methylbut-2-enyl)chromen-4-one
- HY-N4247
- BDBM50540963
- MFCD05662262
- AKOS030573630
- NSC 356888
- AC-34789
- DA-54709
- MS-31140
- 1ST157267
- CS-0032528
- Q15634181
- 4H-1-Benzopyran-4-one, 8-(6-(2,4-dihydroxyphenyl)-3-methyl-2-cyclohexen-1-yl)-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methyl-2-butenyl)-, (1S-(1alpha,5alpha,6beta))-
- 4H-1-Benzopyran-4-one, 8-[(1S,5R,6S)-6-(2,4-dihydroxybenzoyl)-5-(2,4-dihydroxyphenyl)-3-methyl-2-cyclohexen-1-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methyl-2-buten-1-yl)-
- 4H-1-Benzopyran-4-one, 8-[6-(2,4-dihydroxybenzoyl)-5-(2,4-dihydroxyphenyl)-3-methyl-2-cyclohexen-1-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methyl-2-butenyl)-, [1S-(1alpha,5alpha,6beta)]-
- 8-[(1R,2S,3S)-2-(2,4-dihydroxybenzoyl)-2',4'-dihydroxy-5-methyl[1,2,3,6-tetrahydro[1,1'-biphenyl]]-3-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methylbut-2-en-1-yl)-4H-1-benzopyran-4-one
- 8-[(1S,5R,6S)-6-(2,4-Dihydroxybenzoyl)-5-(2,4-dihydroxyphenyl)-3-methyl-2-cyclohexen-1-yl]-2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-3-(3-methyl-2-buten-1-yl)-4H-1-benzopyran-4-one
Property Name
Property Value
Reference
Property Name
Molecular Weight
Property Value
692.7 g/mol
Reference
Computed by PubChem 2.2 (PubChem release 2021.10.14)
Property Name
XLogP3-AA
Property Value
7.3
Reference
Computed by XLogP3 3.0 (PubChem release 2021.10.14)
Property Name
Hydrogen Bond Donor Count
Property Value
8
Reference
Computed by Cactvs 3.4.8.18 (PubChem release 2021.10.14)
Property Name
Hydrogen Bond Acceptor Count
Property Value
11
Reference
Computed by Cactvs 3.4.8.18 (PubChem release 2021.10.14)
Property Name
Rotatable Bond Count
Property Value
7
Reference
Computed by Cactvs 3.4.8.18 (PubChem release 2021.10.14)
Property Name
Exact Mass
Property Value
692.22576196 Da
Reference
Computed by PubChem 2.2 (PubChem release 2021.10.14)
Property Name
Monoisotopic Mass
Property Value
692.22576196 Da
Reference
Computed by PubChem 2.2 (PubChem release 2021.10.14)
Property Name
Topological Polar Surface Area
Property Value
205 Ų
Reference
Computed by Cactvs 3.4.8.18 (PubChem release 2021.10.14)
Property Name
Heavy Atom Count
Property Value
51
Reference
Computed by PubChem
Property Name
Formal Charge
Property Value
0
Reference
Computed by PubChem
Property Name
Complexity
Property Value
1380
Reference
Computed by Cactvs 3.4.8.18 (PubChem release 2021.10.14)
Property Name
Isotope Atom Count
Property Value
0
Reference
Computed by PubChem
Property Name
Defined Atom Stereocenter Count
Property Value
3
Reference
Computed by PubChem
Property Name
Undefined Atom Stereocenter Count
Property Value
0
Reference
Computed by PubChem
Property Name
Defined Bond Stereocenter Count
Property Value
0
Reference
Computed by PubChem
Property Name
Undefined Bond Stereocenter Count
Property Value
0
Reference
Computed by PubChem
Property Name
Covalently-Bonded Unit Count
Property Value
1
Reference
Computed by PubChem
Property Name
Compound Is Canonicalized
Property Value
Yes
Reference
Computed by PubChem (release 2021.10.14)
Follow these links to do a live 2D search or do a live 3D search for this compound, sorted by annotation score. This section is deprecated (see here for details), but these live search links provide equivalent functionality to the table that was previously shown here.
Same Connectivity Count
Same Parent, Connectivity Count
Similar Compounds (2D)
Similar Conformers (3D)
Same Count
The LOTUS Initiative for Open Natural Products Research: frozen dataset union wikidata (with metadata) | DOI:10.5281/zenodo.5794106
- CAS Common ChemistryLICENSEThe data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated.https://creativecommons.org/licenses/by-nc/4.0/
- ChemIDplusChemIDplus Chemical Information Classificationhttps://pubchem.ncbi.nlm.nih.gov/source/ChemIDplus
- EPA DSSToxCompTox Chemicals Dashboard Chemical Listshttps://comptox.epa.gov/dashboard/chemical-lists/
- FDA Global Substance Registration System (GSRS)LICENSEUnless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.https://www.fda.gov/about-fda/about-website/website-policies#linking
- ChEBI
- LOTUS - the natural products occurrence databaseLICENSEThe code for LOTUS is released under the GNU General Public License v3.0.https://lotus.nprod.net/LOTUS Treehttps://lotus.naturalproducts.net/
- ChEMBLLICENSEAccess to the web interface of ChEMBL is made under the EBI's Terms of Use (http://www.ebi.ac.uk/Information/termsofuse.html). The ChEMBL data is made available on a Creative Commons Attribution-Share Alike 3.0 Unported License (http://creativecommons.org/licenses/by-sa/3.0/).http://www.ebi.ac.uk/Information/termsofuse.htmlChEMBL Protein Target Treehttps://www.ebi.ac.uk/chembl/g/#browse/targets
- Comparative Toxicogenomics Database (CTD)LICENSEIt is to be used only for research and educational purposes. Any reproduction or use for commercial purpose is prohibited without the prior express written permission of NC State University.http://ctdbase.org/about/legal.jsp
- Japan Chemical Substance Dictionary (Nikkaji)
- KEGGLICENSEAcademic users may freely use the KEGG website. Non-academic use of KEGG generally requires a commercial licensehttps://www.kegg.jp/kegg/legal.htmlPhytochemical compoundshttp://www.genome.jp/kegg-bin/get_htext?br08003.keg
- Natural Product Activity and Species Source (NPASS)
- Metabolomics Workbench
- PharosLICENSEData accessed from Pharos and TCRD is publicly available from the primary sources listed above. Please respect their individual licenses regarding proper use and redistribution.https://pharos.nih.gov/about
- SpectraBaseMORACENIN-Bhttps://spectrabase.com/spectrum/LAH5mxrTsIO
- Wikidatakuwanone Ghttps://www.wikidata.org/wiki/Q15634181
- Wikipedia
- PubChem
- Medical Subject Headings (MeSH)LICENSEWorks produced by the U.S. government are not subject to copyright protection in the United States. Any such works found on National Library of Medicine (NLM) Web sites may be freely used or reproduced without permission in the U.S.https://www.nlm.nih.gov/copyright.html
- MolGenieMolGenie Organic Chemistry Ontologyhttps://github.com/MolGenie/ontology/
- NCBI
CONTENTS